The Protein Production Puzzle
Stanford University researchers have delved into the intricate processes within our brain cells, uncovering a fundamental molecular issue that may underpin
the aging of the brain. Their groundbreaking work, primarily conducted using the remarkably short-lived turquoise killifish, points to a breakdown in proteostasis – the crucial cellular system responsible for maintaining a delicate balance between creating new proteins and clearing out old or damaged ones. This imbalance, particularly evident in brain cells, appears to pave the way for the accumulation of misfolded proteins, a characteristic hallmark of both normal brain aging and devastating neurodegenerative conditions like Alzheimer's disease. By studying a model organism that ages rapidly, scientists can observe these complex age-related changes much faster, providing invaluable insights into mechanisms that are shared across many species, including humans.
Ribosome Roadblocks Emerge
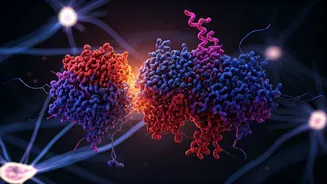
The core of this molecular misadventure, as identified by the Stanford team, lies within a specific stage of protein synthesis known as translation elongation. During this vital process, cellular machinery called ribosomes diligently read genetic blueprints encoded in messenger RNA (mRNA) to assemble proteins, amino acid by amino acid. However, in the brains of older killifish, a significant disruption was observed: ribosomes frequently encountered obstacles, causing them to collide and stall along the mRNA strands. These 'ribosome traffic jams' severely hampered the efficient production of essential proteins and, critically, contributed to the formation of harmful protein aggregates. This discovery highlights how crucial the precise regulation of ribosome speed is for maintaining protein homeostasis, especially as we age.
Untangling Protein-Transcript Decoupling
This new understanding of ribosome dysfunction offers a compelling explanation for a long-standing mystery in aging research: protein-transcript decoupling. This phenomenon refers to the observation that in aging individuals, the levels of certain mRNA molecules no longer accurately reflect the amounts of the proteins they are meant to produce. The Stanford study demonstrates that the compromised protein synthesis process, characterized by stalled ribosomes, is a direct cause of this decoupling. Since many of the proteins affected by these issues are involved in maintaining the integrity and proper functioning of our genetic material (genome maintenance), this finding sheds light on why these critical processes also tend to falter with age. Essentially, problems in the protein factory itself lead to wider systemic failures.
Therapeutic Avenues Beckon
The implications of these findings extend beyond simply understanding the aging process; they open promising avenues for therapeutic intervention. By identifying ribosome dysfunction as a key culprit in the decline of proteostasis, researchers are now exploring whether targeting translation efficiency or improving ribosome quality control mechanisms could offer a way to restore healthy protein production in brain cells. Such interventions might not only combat the detrimental effects of aging but could potentially delay age-related cognitive decline and even mitigate the progression of neurodegenerative disorders. The research team is eager to further investigate how these ribosome-related issues contribute to age-related brain diseases in humans and to assess the feasibility of developing treatments to enhance brain cell resilience.