Prescription Mandate Issued
The Central Drugs Standard Control Organisation (CDSCO), India's primary drug regulatory body, has issued a significant advisory concerning the upcoming
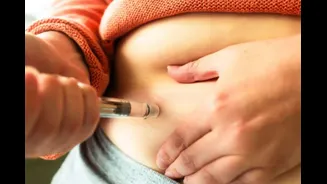
influx of generic weight loss drugs, specifically mentioning GLP-1 receptor agonists like semaglutide. Ahead of their anticipated market entry, the CDSCO has stipulated that these medications must be sold exclusively upon the presentation of a valid prescription from a registered medical practitioner. This directive is a proactive step to manage the sale and distribution of these potent pharmaceuticals, ensuring they are used under appropriate medical supervision and not subjected to indiscriminate public access. The move comes as the patent expiry for blockbuster drugs is expected to lead to a surge in generic alternatives, with approximately 50 brands poised for launch in the near future. The regulatory body's firm stance aims to prevent any potential misuse or over-reliance on these drugs for weight management without professional medical guidance.
Combating Misleading Marketing
The advisory from the CDSCO explicitly targets and prohibits any promotional activities designed to mislead the public or inflate demand for these prescription drugs. Activities such as creating unwarranted demand, exaggerating the drug's effectiveness, or downplaying the critical role of lifestyle modifications in weight management are classified as deceptive marketing practices. The regulatory body has observed that some pharmaceutical companies might be employing various indirect or surrogate promotional strategies. These can include disease awareness campaigns, digital outreach, and other communications that, while seemingly educational, serve to enhance brand recall and product visibility for prescription-only medicines. The CDSCO views such tactics, including those under the guise of influencer engagement or corporate campaigns, with serious concern, categorizing them as irrational or misleading marketing, and warns of stringent actions against violations.
Ensuring Safe Market Entry
The GLP-1 receptor agonists, initially approved for Type 2 diabetes treatment in 2017, gained widespread recognition for their efficacy in weight loss, with a specific higher-dose version gaining FDA approval for this purpose in 2021. The global surge in popularity, fueled partly by celebrity endorsements, saw many individuals in India seeking access to these drugs even before their official launch in the country. Both semaglutide and a successor drug entered the Indian market last year. With a substantial number of generic versions set to launch soon, the regulator has urged all manufacturers, importers, and market authorization holders to rigorously adhere to the provisions of the Drugs and Cosmetics Act. Furthermore, package inserts are required to include a dedicated contact number for consumer queries and a complaint mechanism, fostering transparency and accountability. Companies are also tasked with submitting comprehensive risk management plans to ensure continuous monitoring and the implementation of effective risk minimization strategies, prioritizing the protection of vulnerable populations from exploitation.