The Rise of Semaglutide
The burgeoning market for weight management solutions in India is experiencing an unprecedented surge, primarily fueled by medications containing semaglutide.
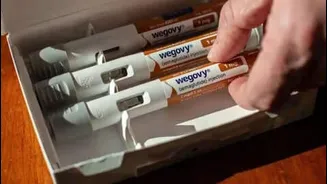
While the branded drug Ozempic, known for its efficacy, isn't officially sanctioned for weight loss purposes in India, its off-label application and the rapid emergence of generic alternatives have cultivated a substantial demand. This phenomenon is further propelled by increasing awareness among the populace regarding the effectiveness of these treatments for shedding excess weight, a trend amplified by celebrity endorsements and broader public discourse. The availability of these medications, irrespective of the channels through which they are obtained, underscores a significant shift in how individuals are approaching weight management, moving towards pharmacological interventions as a key component of their strategy.
Affordable Alternatives Emerge
The impending expiration of the patent for semaglutide, a widely recognized drug for both diabetes and weight management, is poised to dramatically reshape the Indian pharmaceutical landscape. With the patent set to lapse on March 20, an estimated 50 generic brands are anticipated to flood the market shortly thereafter. This influx is expected to not only broaden access to this class of innovative medicines but also lead to a substantial reduction in costs. Industry experts predict that the monthly expenditure for once-a-week semaglutide injectables could decrease significantly, potentially falling between Rs 3,000 and Rs 4,000. This is a stark contrast to the current pricing of branded versions like Ozempic, which range from Rs 8,800 to Rs 11,175 monthly, and Wegovy, costing between Rs 10,850 and Rs 16,400. These generic versions, being bioequivalent to their branded counterparts in terms of dosage, safety, strength, quality, and performance, offer the same therapeutic benefits at a considerably lower price point, as they bypass the extensive and costly clinical trials required for original drug development.
Understanding Semaglutide's Appeal
The considerable appeal of semaglutide stems from its sophisticated mechanism of action, which effectively mimics the body's natural GLP-1 hormone. This hormone, typically released post-meal, plays a crucial role in regulating blood sugar by stimulating insulin production, slowing down digestion, and importantly, suppressing appetite. Initially approved by the US FDA in 2017 for managing Type-2 diabetes, its profound effect on reducing hunger quickly garnered attention for its potential in weight management. This led to the US FDA's approval of a higher-dose version specifically for weight loss in 2021. Novo Nordisk, the original developer, marketed Ozempic primarily for diabetes, but its appetite-suppressing capabilities made it a sought-after weight loss agent. Their specialized weight-loss formulation, Wegovy, was introduced in India last year, prior to the patent's expiry, further solidifying semaglutide's prominent position in the market.
Market Dynamics and Competition
The Indian market is gearing up for a significant transformation with the launch of nearly 50 semaglutide brands. However, market analysts anticipate that only a handful of larger pharmaceutical players are likely to capture substantial market share, similar to the trends observed with other off-patent diabetes medications. Major companies such as Dr Reddy's, Zydus, Sun Pharmaceuticals, Cipla, and Lupin are poised to enter the fray, with some, like Zydus Lifesciences, planning to introduce multiple brands such as Semaglyn, Mashema, and Alterme. Zydus is also innovating with multi-dose semaglutide pens, designed to simplify treatment by allowing patients to adjust dosages, which is particularly beneficial during the gradual dose-escalation phase, thereby potentially improving long-term patient adherence. Despite the increased competition, the leading innovator companies are not expected to significantly alter their current pricing strategies, emphasizing the robust clinical evidence and quality of their established products.
Navigating Risks and Regulations
The widespread availability and increased marketing of these potent weight loss drugs also bring inherent risks, including potential misuse and the need for strict medical supervision. Concerns have been raised about the aggressive marketing tactics employed by some manufacturers, leading the Central Drugs Standard Control Organisation (CDSCO) to issue advisories warning against promotional activities that circumvent regulations. Furthermore, pharmacist associations are reinforcing the importance of dispensing these medications strictly based on a doctor's prescription, especially given anecdotal reports of off-label sales. Medical professionals emphasize that while these drugs can be highly effective for medically supervised obesity management, they are not a panacea and should be integrated with lifestyle changes. Adverse effects, ranging from gastrointestinal issues to more serious conditions like pancreatitis and thyroid concerns, necessitate careful monitoring by healthcare providers to ensure patient safety and responsible use.