Dramatic Price Drop
The landscape of weight management and Type 2 diabetes treatment in India has been dramatically reshaped with the recent market entry of generic semaglutide.
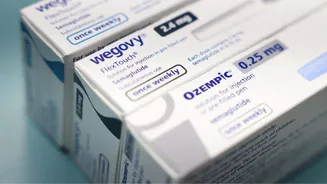
Following the expiration of the originator drug's patent, domestic pharmaceutical giants have unleashed their versions, leading to a staggering price reduction of 70-90%. This substantial decrease means that the monthly cost for the lowest dosage of this revolutionary GLP-1 receptor agonist can now be as low as Rs 1,290. This makes a once prohibitively expensive treatment accessible to a much broader segment of the population, signaling a significant shift in healthcare affordability. Analysts predict a doubling of GLP-1 unit sales within the next three months due to these steep price cuts, indicating strong patient and physician adoption of these more economical options. The original branded medications, like Ozempic and Wegovy, which previously ranged from Rs 8,800 to Rs 16,400 per month, are now facing intense competition from these new, budget-friendly alternatives.
Key Market Players
Several major Indian pharmaceutical companies are now vying for market share with their generic semaglutide offerings, each bringing unique formulations and pricing strategies. Two prominent players, Natco Pharmaceuticals and Eris Lifesciences, have set the benchmark for affordability, launching their lowest monthly doses at Rs 1,290. Natco will offer its products, 'Semanat' and 'Semafull,' in 2 mg, 4 mg, and 8 mg doses. Eris Lifesciences, in a strategic partnership with Natco, will introduce its version under similar pricing structures. Glenmark Pharmaceuticals has launched GLIPIQ®, available in both vial and pre-filled pen formats, with weekly treatment costs ranging from Rs 325 to Rs 440 (Rs 1,300 to Rs 1,760 monthly). Zydus Lifesciences offers its generic semaglutide under brand names like Semaglyn and Mashema, with an average monthly cost around Rs 2,200, and importantly, includes a reusable multi-dose pen device allowing for dose titration. Sun Pharma has introduced Noveltreat and Sematrinity, priced competitively, with weekly therapies for Noveltreat spanning Rs 900 to Rs 2,000, and for Sematrinity from Rs 750 to Rs 1,300. Dr Reddy's Laboratories, an early entrant with DCGI approval, markets its injectable under the brand Obeda®, available in 2 mg and 4 mg strengths via pre-filled pens at a monthly cost of Rs 4,200 for both strengths.
Formats and Patient Support
The introduction of generic semaglutide is not just about price; it's also about enhancing patient convenience and support. While many companies are offering traditional vial formats, pre-filled pens are becoming increasingly popular for their ease of use and accurate dosing, especially for self-administration once a week. Glenmark's GLIPIQ® comes in both vial and convenient pre-filled pen options, catering to different patient preferences. Natco plans to launch an easy-to-inject pen device next month, complementing their vial offerings. Zydus is notable for its reusable multi-dose pen device, allowing patients to adjust their dosage, a significant advantage over single-dose pens. Sun Pharma and Glenmark are going a step further by implementing comprehensive patient guidance and support programs. These initiatives aim to help patients navigate their treatment journey, from initial therapy initiation to long-term adherence, and address any anxieties associated with injectable medications. Sun Pharma's Noveltreat features a concealed needle for improved safety and handling, while Sematrinity offers a multi-dose pen with a smooth dialer for precise delivery. Glenmark's 'Sankalp' program is specifically designed to ease therapy initiation and build comfort with the treatment regimen.
Market Impact and Future Outlook
The entry of affordable generic semaglutide is poised to significantly expand the GLP-1 drug market in India. Currently, the market is valued at approximately Rs 1,500 crore, with Eli Lilly's Mounjaro dominating at 85%, and Novo Nordisk's Ozempic and Wegovy holding the remainder. With the patent expiry and price reductions, analysts anticipate a two-fold increase in GLP-1 unit sales over the next three months. This surge is driven by greater accessibility for obesity management, a chronic condition requiring long-term treatment, and the shift towards more cost-effective semaglutide options. Experts predict that over 43 companies will eventually enter the market with generic versions, creating a competitive environment that maintains affordability. While a price war was initially anticipated, the generics are expected to settle within a narrow, substantially lower price band compared to the innovator products. This development democratizes access to vital treatments, offering hope for millions struggling with obesity and Type 2 diabetes to manage their health more effectively and affordably.