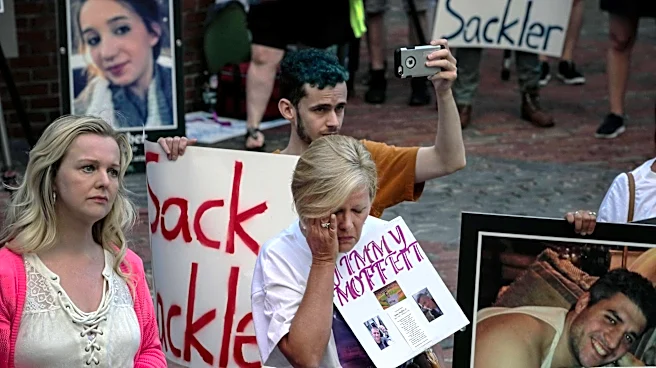By Dietrich Knauth
April 24 (Reuters) - Tammy Blanton’s life unraveled after years of taking opioids initially prescribed for migraines, according to her daughter Mary Anne, who says the drugs left her mother
isolated, unemployed and estranged from her family.
Tammy was given opioid prescriptions by multiple providers for decades — receiving more than 200 pills a month on average during one two-year period — and a medical examiner later concluded that oxycodone and extended‑release morphine, along with alcohol and anti-anxiety drugs, contributed to her accidental death at age 58 in 2017.
When Purdue Pharma sought bankruptcy protection in 2019, Blanton believed her mother’s story would qualify her for compensation. Purdue, whose painkiller OxyContin has been widely blamed for fueling the opioid crisis, acknowledged misconduct and pledged to compensate those harmed.
While lawsuits over the crisis have generated more than $57 billion in settlements — mostly pledged to state and local governments — Purdue’s deal is the only major agreement to set aside a substantial sum for individuals harmed by opioids, with about $865 million earmarked for them.
The fund represents the last and best chance for victims of the opioid crisis to receive any compensation. The sprawling litigation that once targeted nearly every major opioid manufacturer, distributor and pharmacy chain is largely over, and no comparable fund for individuals is coming.
For Blanton and many others, that promise is now slipping away. Reuters analyzed the vast bankruptcy record built over six years, including hundreds of legal filings, more than 100 letters from people seeking compensation, and interviews with eight victims and lawyers close to the case. The deal was hailed by both the company and the plaintiffs' lawyers as a victory for victims, but the news agency’s examination shows how the long, grinding bankruptcy wound up creating daunting hurdles for many trying to qualify for compensation.
Blanton is among those who may receive nothing from the deal because they cannot produce records proving Purdue — and not a generic competitor — made the pills they or their relatives were prescribed. Many individuals were able to file claims without documentation when the process began, only to learn years later that records they needed had been destroyed.
The requirement that victims prove they took a Purdue-manufactured opioid can be very difficult to meet years later. Doctors' records typically list the drug prescribed, not the manufacturer. Insurance companies often steer patients toward generics to save money. Pharmacies can switch suppliers over time, and in many states, neither they nor doctors, hospitals nor insurers are required to retain records for more than a few years.
“To me, it's irrelevant whether Purdue manufactured her specific prescription — it ultimately came from them,” Blanton said. Purdue told “everybody that they were safe and not addictive. They created this mess.”
Members of the Sackler family, which owned Purdue, referred to the company for comment. Purdue declined multiple requests for comment.
BILLIONS, BUT FOR WHOM?
The documentation requirement was embedded in the bankruptcy plan Purdue negotiated with its creditors, reflecting its long-standing position that it should be held liable only for harms directly traceable to its products.
Victims and their lawyers say Purdue and its Sackler family owners should be held broadly responsible for igniting an opioid epidemic through aggressive and misleading marketing that drove widespread use of prescription painkillers, including generics, with many patients later turning to illegal drugs.
Purdue has twice pleaded guilty to federal criminal charges related to its marketing of OxyContin, admitting it misled regulators, doctors and patients about addiction risks and engaged in illegal practices to boost opioid sales.
When the company filed for Chapter 11, people who said they had been harmed by its opioid pills became creditors in its case — placed in the same legal category as states, cities and other governments that had sued the company.
In March 2021, when Purdue introduced its initial bankruptcy plan, Steve Miller, chairman of the company’s board of directors, called it “historic” and said it would have “a profoundly positive impact on public health by directing critically‑needed resources to communities and individuals nationwide.”
Purdue encouraged individuals to file claims. Almost 140,000 people did so by the September 2021 deadline, filling out a seven‑page form that did not require detailed documentation. Some claims were filed by lawyers representing many clients, but many were submitted by people struggling with addiction or unable to afford a lawyer.
Purdue’s bankruptcy then dragged on for years, becoming entangled in appeals that eventually reached the U.S. Supreme Court. The settlement was negotiated behind closed doors, through confidential mediation sessions, as part of a complex bankruptcy that produced more than 9,000 court filings.
In May 2025, nearly four years after the deadline for filing claims had passed, a trustee appointed to administer the fund requested for the first time that people produce records proving that Purdue had manufactured the drug that caused their harm. He set a deadline of 60 days. The long delay made it more likely that the requested documents would no longer be available from doctors, pharmacies or insurers, which generally only need to retain records for a few years.
An earlier version of the plan would have allowed people without prescription records to qualify for a $3,500 payment if they signed a sworn affidavit saying they had used the drug. People with records and more serious harms could qualify for up to $48,000, according to court filings. But after the appeals process, the reworked deal limited payouts only to people who had records. The change was not discussed openly in court, which ProPublica was the first to report in a piece published Thursday.
Ed Neiger, an attorney who helped represent a group of about 30,000 victims, said plaintiffs’ lawyers sought to make the requirements about what proof individuals needed to provide as flexible as possible, but ran up against demands from other lawyers negotiating the bankruptcy settlement that claimants provide proof similar to what would be required in a lawsuit. ”We couldn’t get it to a point where you could get recovery without a prescription. And the option was either, you know, blow up the settlement or take the concessions that you were able to extract.”
Despite its shortcomings, the Purdue settlement offers an easier path to compensation than traditional litigation, Neiger said. Individual lawsuits against Purdue or the Sackler family would likely have taken years, cost significant sums and required far more detailed proof, with no assurance of success. No individual has ever successfully sued the Sacklers or Purdue over a personal opioid addiction.
Nevertheless, more than 40% of the claims filed have already been rejected by U.S. District Judge Sean Lane in White Plains, New York, who is overseeing Purdue’s bankruptcy.
Even for those whose claims will be approved, recoveries are expected to be relatively modest. Purdue estimated in December that eligible individuals could receive about $8,000 or $16,000, depending on how long opioids were prescribed. Those figures are estimates, and they could rise if fewer claimants ultimately meet the documentation requirements, since the pool of money would be divided among fewer people.
PAPER TRAIL, DEAD ENDS
Purdue sold extended-release morphine under the name MS Contin and later extended-release oxycodone under the name OxyContin — the same drugs Blanton said her mother took in vast quantities over decades. She said in an interview that she knows her mother took some Purdue-manufactured pills, but could not prove that Purdue made the morphine or oxycodone her mother took. Purdue developed and first marketed both drugs, but many other companies later won approval to sell generic versions.
After her mother’s death, Blanton began searching for records from doctors, hospitals and pharmacies to support her claim, but said much of the information required either no longer exists or was never recorded in the first place. Tammy’s primary care physician had legally destroyed her records, Blanton said, and hospital records she obtained often did not identify the manufacturer. She was also unable to obtain records from Arizona’s Medicaid program, which paid for most of her mother’s prescription, because of documentation hurdles tied to proof of next‑of‑kin status and privacy rules.
Purdue says it has taken a flexible approach to documentation, accepting a range of evidence including prescription records, references to Purdue opioids in other qualifying documents, or photographs of prescription bottles. In a January court filing, the company described its requirements as “flexible and far less onerous” than the proof a plaintiff would need in a lawsuit.
Michele Capozzi‑Pollock, a 59-year-old Massachusetts resident whose husband died after years of opioid use, laughed when she was told pill bottles could be used as proof of a claim. “Like I’m going to save 16 years’ worth of prescription bottles,” she said in an interview.
Capozzi-Pollock said she was told the claim would be denied because she did not respond to the documentation request sent last summer and addressed to her husband three years after his death.
“How much time do I put towards this, and energy and money, just to get to the end and then have them say, ‘No, denied’?”
“AT A LOSS”
When Purdue in January asked the bankruptcy court to expunge more than 57,000 claims from people who did not respond to the trustee’s May 2025 request for documentation, hundreds of victims sent letters to the court in protest.
Their letters describe not only difficulty obtaining records, but basic confusion about how the settlement works.
“I am at a loss and do not know what to do,” wrote Terry Hughes, an inmate at Huttonsville Correctional Center in West Virginia, in a letter to the bankruptcy court dated February 20. Hughes said the pharmacy where he filled opioid prescriptions had closed years ago.
Michael Galipeau, a 41-year-old resident of Red Hook, New York, received an email in January with the subject line: “Purdue Pharma L.P., et al., Case No. 19-23649 Omnibus Claims Objection to Unsubstantiated Claims,” almost six years after filing his claim.
Galipeau, who has struggled with opioid addiction for nearly two decades, became dependent on painkillers prescribed for a broken wrist in 2007, served time in prison for dealing drugs, and now counsels people recovering from addiction.
It wasn’t until he reached page 3,024 of a 17,101-page PDF attachment that he saw the words: “Claimant failed to provide information to substantiate claim.”
In an interview, Galipeau, who attended a February 26 court hearing in White Plains, said he tried to argue to Lane, the judge overseeing the case, that the settlement’s documentation requirements were too restrictive. Lane cut him off and moved on to other speakers, including dozens who joined by Zoom.
During the hearing, the judge acknowledged the frustration many people expressed — over bureaucratic complexity and a sense that the process had left ordinary victims without guidance or recourse. Still, he agreed to Purdue's request to dismiss nearly all of the 57,000 claims.
Lane declined to comment.
The hearing, which Reuters listened to by phone, was one of several scheduled for this spring and summer as the court considers whether to dismiss the tens of thousands of remaining claims.
Not everyone will be shut out. Jill Cichowicz, a 47‑year‑old Richmond, Virginia resident who lost her twin brother Scott to an overdose in 2017, said she has records showing he was prescribed OxyContin and expects to qualify for a payment. She said Scott kept meticulous notes about the drugs he was taking and that her family hired an investigator and saved pill bottles listing Purdue as the manufacturer after his death — advantages she said many families lack. “I don't think the average person that's battling addiction is keeping, you know, copious records and Excel spreadsheets of everything they're taking,” Cichowicz said. “I think they're just trying to survive.”
(Reporting by Dietrich Knauth, in New York; Editing by Alexia Garamfalvi, Amy Stevens and Claudia Parsons)