What's Happening?
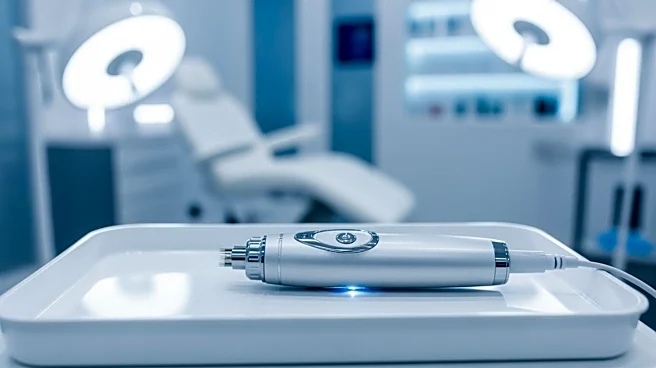
Dermalogica has introduced the PRO Pen Microneedling System, marking its first medical device cleared by the United States Food and Drug Administration (FDA). This development represents a significant step for the professional skincare brand as it expands
further into advanced aesthetics. The PRO Pen is designed for licensed professionals and features dual functionality for microneedling, a treatment that stimulates collagen production by creating controlled microinjuries in the skin. Additionally, it includes Luminfusion, an exfoliation and infusion treatment that combines nanoneedling, active ingredients, and LED red light therapy. Dermalogica emphasizes minimal downtime and expert oversight as key differentiators in a crowded market. The company, owned by Unilever, is focusing on maintaining advanced treatments in qualified hands amidst a surge of consumer-facing devices and minimally invasive treatments.
Why It's Important?
The introduction of the PRO Pen Microneedling System is significant as it underscores Dermalogica's commitment to the professional skincare channel, which is a core part of its business model. The FDA clearance provides reassurance to both skin therapists and clients, ensuring safety and performance standards are met. This move comes at a time when the microneedling market is projected to grow significantly, with Mordor Intelligence estimating a 10.9% compound annual growth rate, reaching $1.1 billion by 2030. The launch also highlights a broader trend in the aesthetics industry, where treatments like microneedling, lasers, and injectables are becoming mainstream. Dermalogica's focus on integrating skincare and aesthetics presents an opportunity to enhance long-term skin health, distinguishing itself from quick cosmetic fixes.
What's Next?
Dermalogica's focus on the professional channel suggests a continued investment in training and supporting licensed skin therapists. The company trains over 75,000 skin therapists annually and views the professional channel as the engine of its past and future growth. As the aesthetics industry evolves, Dermalogica aims to maintain its emphasis on long-term skin health rather than structural cosmetic enhancements. The company plans to leverage its FDA-cleared device to strengthen its position in the professional skincare market, ensuring that advanced treatments remain in the hands of qualified professionals. This approach may influence other brands to prioritize professional expertise amidst the rise of AI-driven diagnostics and personalization tools.
Beyond the Headlines
The launch of the PRO Pen Microneedling System reflects a broader shift in the beauty industry towards personalized and holistic skincare solutions. As consumers increasingly seek treatments that address long-term skin health, Dermalogica's strategy aligns with this demand by emphasizing the importance of professional expertise and human relationships. The company's stance against quick cosmetic fixes and its focus on maintaining skin integrity over time may set a precedent for other brands in the industry. Additionally, the integration of AI in skincare diagnostics highlights the need for a balanced approach that combines technology with professional judgment.