What's Happening?
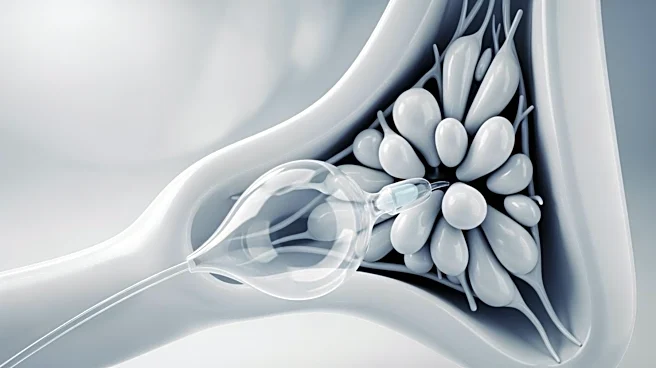
Corvention, a medical device company based in Flagstaff, Arizona, has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its KardiaPSI Balloon Catheter. This device is designed for balloon aortic valvuloplasty (BAV), offering
an ultra high-pressure solution for the precise dilation of stenotic aortic valves. The KardiaPSI Balloon Catheter features a 24 atm pressure capability within a compact 2.5 cm balloon, aimed at addressing the mechanical challenges posed by calcified valve anatomy. The device promises enhanced predictability and accuracy during valvuloplasty procedures, which are critical for patients with calcified aortic valves. Corvention plans a controlled, phased market introduction to ensure high standards of clinical support and physician training.
Why It's Important?
The FDA clearance of the KardiaPSI Balloon Catheter is significant as it introduces a new tool for interventionalists dealing with complex aortic valve procedures. The device's high-pressure capability and precision design could improve patient outcomes by providing more reliable and controlled dilation of calcified valves. This advancement is particularly important for patients with severe aortic stenosis, a condition that can lead to heart failure if not properly managed. By enhancing the tools available for such procedures, Corvention is contributing to the broader field of structural heart interventions, potentially setting new standards for safety and efficacy in cardiovascular treatments.
What's Next?
Following the FDA clearance, Corvention is preparing for a national rollout of the KardiaPSI Balloon Catheter. The company is focusing on establishing the necessary commercial infrastructure and strategic support networks to ensure a successful launch. This includes prioritizing clinical support and physician training to maximize the device's impact in clinical settings. As the device enters the market, it will be crucial to monitor its adoption and effectiveness in real-world applications, which could influence future developments in the field of structural heart interventions.