What's Happening?
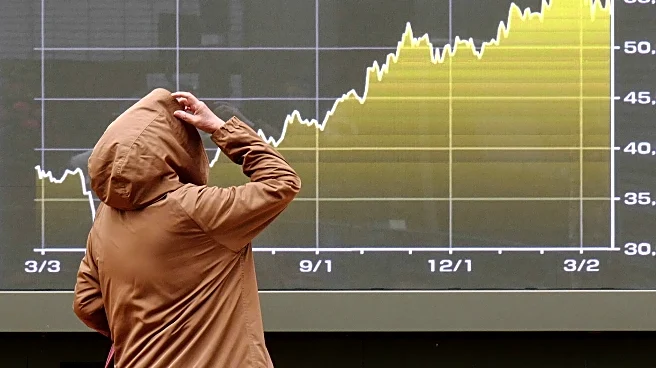
Abivax, a French biotech company, is preparing to release key trial data in June, which its CEO, Marc de Garidel, believes will secure better terms for future partnerships. The company has been the subject of intense takeover rumors, with its stock rising
nearly 1,700% in 2025. Analysts view Abivax as a prime takeover target, with several pharmaceutical giants rumored as potential buyers. The upcoming trial will assess the long-term maintenance effect of Abivax's lead asset, obefazimod, which is seen as a potentially best-in-class treatment for ulcerative colitis and is also being tested for Crohn's disease. The company plans to apply for U.S. Food and Drug Administration approval in the fourth quarter, contingent on positive trial results.
Why It's Important?
The outcome of the June trial is critical for Abivax as it could significantly influence the company's valuation and attractiveness to potential buyers. Positive results could open up a multi-billion-dollar market for treatments of Irritable Bowel Syndrome (IBS), enhancing Abivax's negotiating power in partnership discussions. The company's strategic decision to wait for trial results before entering into deals suggests confidence in the drug's potential and a focus on maximizing shareholder value. This development is closely watched by investors and industry stakeholders, as it could lead to significant shifts in the biotech market landscape.
What's Next?
Following the trial results, Abivax plans to raise funds through a combination of equity financing and debt to support its path to profitability. The company has indicated it has sufficient cash to operate until late 2027, with a reported cash reserve of 530 million euros as of the end of 2025. The anticipated fundraising will likely occur in late June, with the amount expected to be several million euros. The trial results and subsequent financial strategies will be pivotal in determining Abivax's future, including potential partnerships or acquisitions.