What's Happening?
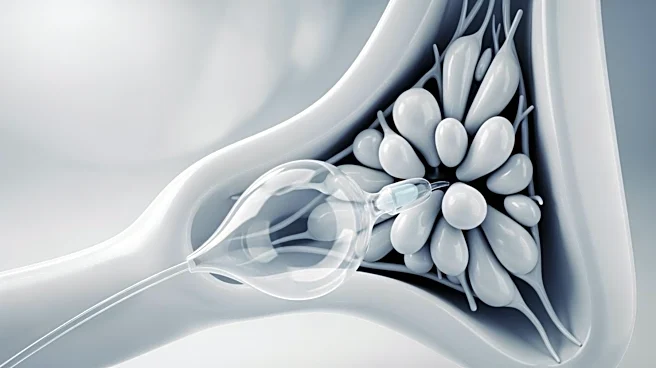
Anteris Technologies Global has commenced US patient enrolment in its pivotal PARADIGM trial for the DurAVR Transcatheter Heart Valve (THV) system. The company has secured substantial funding, including a public offering and a private placement with Medtronic,
raising a total of US$320 million. This financial boost has increased Anteris's cash reserves to US$283.2 million as of March 31, 2026. The PARADIGM trial, targeting 1,000 patients, aims to compare DurAVR against commercially available TAVRs. The trial's progress is supported by US Medicare reimbursement eligibility, facilitating patient recruitment and site-level adoption.
Why It's Important?
The initiation of the PARADIGM trial marks a significant milestone for Anteris Technologies in the development of its DurAVR THV system, which could offer a new treatment option for patients requiring transcatheter aortic valve replacement. The substantial funding secured by the company underscores investor confidence and provides the necessary resources to advance the trial and manufacturing scale-up. The trial's success and subsequent regulatory approval could position Anteris as a key player in the cardiovascular device market, potentially improving patient outcomes and expanding treatment options.
What's Next?
Anteris will continue to focus on patient enrolment and advancing the PARADIGM trial, with the goal of achieving regulatory approval for the DurAVR THV system. The company will also work on strengthening its operational infrastructure and manufacturing capabilities, supported by its recent ISO 13485 certification. The success of the trial and regulatory approval will be critical for future commercialization and market entry. Anteris must manage its cash burn and ensure patient recruitment meets expectations to mitigate financing risks.