Affordable Access Unlocked
The pharmaceutical landscape in India has recently undergone a significant transformation, particularly for individuals managing diabetes and weight concerns.
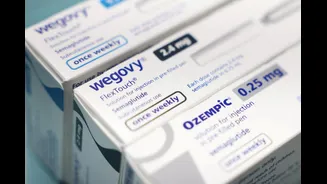
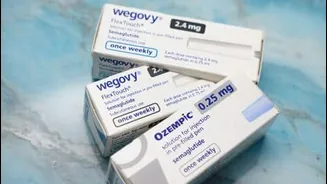
Following the expiration of a crucial patent on March 20th, domestic drug manufacturers are now free to produce and market their own versions of semaglutide. This development is set to dramatically lower treatment costs, moving from an approximate ₹11,000 monthly expense for branded versions to an estimated ₹3,000 to ₹5,000. Projections indicate that as more companies enter the market, prices could descend even further, potentially reaching as low as ₹1,500 to ₹2,500. This price reduction of 50 to 70 percent is a game-changer, significantly enhancing the availability of this vital medication within India's predominantly out-of-pocket healthcare system. Companies like Zydus Lifesciences have already introduced a semaglutide injection in a multi-dose pen, specifically indicated for Type 2 diabetes mellitus and obesity, with a monthly treatment cost around ₹2,200. The market is expected to see a surge of new brands, with over 40 firms anticipated to launch their products, intensifying competition and broadening patient access.
Global Ambitions Brew
The impact of semaglutide's patent expiry in India extends beyond domestic borders. Several Indian pharmaceutical companies are not only focused on meeting the growing local demand but are also strategically planning for global expansion. Dr Reddy’s Laboratories, for instance, has confirmed that the semaglutide it is bringing to the Indian market is the same formulation it supplies internationally. The company possesses the manufacturing capabilities to cater to both domestic needs and export markets, and is actively pursuing regulatory approvals in other countries, with discussions already underway for a potential launch in Canada. Industry experts anticipate that this influx of affordable semaglutide, coupled with increased patient uptake, could elevate India's GLP-1 market to approximately $1 billion within the next few years. This international outlook signifies India's growing prowess in pharmaceutical manufacturing and its potential to become a major global supplier of essential medications.
Semaglutide's Multifaceted Role
Semaglutide, a medication primarily recognized for its dual application in managing Type 2 diabetes and aiding in weight loss, operates by mimicking the action of a naturally occurring hormone called GLP-1. This mechanism leads to a cascade of beneficial effects: it stimulates the body to release insulin, curtails glucose production in the liver, slows down the rate at which food empties from the stomach, and crucially, helps suppress appetite. The combined impact of these actions is instrumental in achieving better blood sugar control and facilitating significant weight reduction, with clinical trials demonstrating reductions in blood sugar and weight loss averaging between 10 to 15 percent or more. Beyond its primary therapeutic uses, semaglutide offers additional cardiovascular benefits, with studies like SUSTAIN-6 showing a notable decrease in the risk of heart attack by 26 percent and stroke by 39 percent. Emerging research also points to potential advantages for kidney and liver health, leading many physicians to view semaglutide as a transformative therapy that actively modifies disease progression rather than merely managing symptoms.
Access vs. Misuse Concerns
The substantial reduction in semaglutide prices is poised to dramatically improve access for a vast number of patients in India who previously found the treatment prohibitively expensive. This increased affordability is a welcome development, especially for those seeking effective management of diabetes and obesity. However, this positive stride is accompanied by growing concerns among medical professionals regarding the potential for misuse. There's a recognized risk that semaglutide might be sought for purely cosmetic weight loss or used without proper medical oversight. As a prescription-only medication, semaglutide requires stringent medical supervision, including careful dose adjustments and integration with dietary management, to ensure safety and efficacy. While common side effects like nausea are generally manageable, rarer but serious risks such as pancreatitis, gallbladder issues, and kidney complications necessitate professional monitoring. Discussions are also ongoing regarding the nuances between original biologic drugs and their generic counterparts; however, it's important to note that approved generic versions must adhere to rigorous regulatory standards for safety and effectiveness.