Localized Treatment, Global Impact
A pioneering study has unveiled a transformative cancer therapy where a single injection of a specially engineered antibody, known as 2141-V11, administered
directly into a tumor, has yielded astonishing results. This localized intervention has proven capable of not only shrinking the treated tumor but also initiating a systemic immune response that attacks cancerous growths across the entire body. In a small phase 1 trial involving 12 patients with advanced cancers like melanoma, renal cell carcinoma, and aggressive breast cancers, six individuals experienced tumor shrinkage. Astonishingly, two patients achieved complete remission, with all traces of cancer disappearing. This breakthrough suggests that a targeted, localized treatment can effectively mobilize the body's own defenses to combat metastatic disease, opening a new and exciting chapter in cancer care by demonstrating a profound systemic effect from a local therapeutic approach.
Unlocking Immune Power
For over two decades, researchers have explored CD40 agonist antibodies as potential cancer fighters by targeting the CD40 receptor on immune cells to boost anti-tumor immunity. However, earlier attempts were hampered by significant toxicities, including widespread inflammation and organ damage, even at low doses. A significant leap forward occurred in 2018 when Jeffrey V. Ravetch and his team at Rockefeller University redesigned the antibody. Their engineered version, 2141-V11, was modified to improve its interaction with a specific Fc receptor, enabling more efficient activation of immune cells against tumors while dramatically reducing severe systemic side effects. Pre-clinical studies in mice had already hinted at this improved safety and efficacy through direct tumor injection, suggesting a more controlled and potent activation of the immune system.
Remarkable Remission Stories
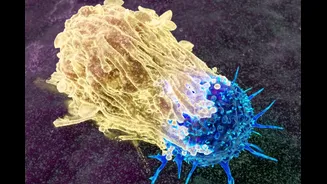
The transformative potential of this new therapy was vividly illustrated by the experiences of two patients. One individual battling metastatic melanoma, who had numerous tumors spread across her leg and foot, saw all her growths vanish after a single tumor on her thigh was injected. Similarly, a patient with metastatic breast cancer experienced the complete disappearance of all tumors, including those in her liver, lungs, and skin, following the injection of just one skin tumor. Post-treatment analysis of non-injected tumors revealed the presence of tumor-infiltrating lymphocytes (TLS), confirming that the immune system had been successfully activated and dispatched to distant cancer sites. This indicates that once the immune system recognizes cancer cells, it can effectively migrate and engage them throughout the body, a critical mechanism driving these extraordinary outcomes.
Expanding Trials, Future Prospects
Buoyed by these exceptional early results, the research team has expanded their trials to include approximately 200 patients across prominent institutions like Memorial Sloan Kettering, Duke University, and Rockefeller University. The ongoing phase 1 and 2 studies are designed to identify patient subgroups most likely to benefit from this treatment and to optimize its administration and dosage. Initial observations suggest that patients who achieved complete remission possessed higher levels of T-cell proliferation at the outset, hinting that a pre-existing immune readiness might be a key factor for successful treatment. Understanding these predictive indicators is crucial for refining the therapy and potentially enabling researchers to convert non-responders into responders, a significant challenge in current immunotherapy treatments where response rates typically range from 25% to 30%.