The Generic Surge
The pharmaceutical landscape in India is set for a major shift with the upcoming expiration of patents on key weight loss drugs, particularly those containing
semaglutide. This event is poised to unleash a wave of generic alternatives, dramatically reducing the cost of these treatments. While branded versions like Ozempic and Wegovy have been available, their premium pricing limited accessibility. However, with approximately 50 generic brands slated for launch following the patent expiry on March 20, patients can anticipate a significant drop in monthly expenses, potentially ranging from Rs 3,000 to Rs 4,000. This contrasts sharply with the current costs of branded semaglutide, which can range from Rs 8,800 to Rs 16,400 per month, depending on the specific drug and dosage. These new generic versions are designed to be bioequivalent to their branded counterparts, offering the same dosage, safety, quality, and performance, but at a fraction of the cost, as they bypass the need for extensive and expensive clinical trials. This development is expected to democratize access to advanced weight management therapies in India.
Semaglutide's Appeal Explained
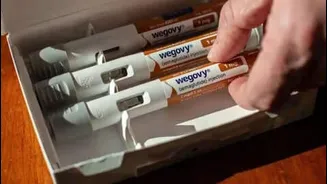
The immense popularity of semaglutide stems from its unique mechanism of action, which effectively mimics the body's natural GLP-1 hormone. This hormone, released in the gut after a meal, plays a crucial role in regulating blood sugar by stimulating insulin production, slowing down digestion, and significantly reducing appetite. Originally approved by the US FDA in 2017 for Type-2 diabetes management, semaglutide quickly garnered attention for its potent weight-loss properties when used off-label. Recognizing this efficacy, a higher-dose formulation specifically for weight management received FDA approval in 2021. The primary developer, a Danish pharmaceutical company, initially marketed Ozempic for diabetes, but its appetite-suppressing capabilities made it a sought-after weight loss agent. Subsequently, a specialized version, Wegovy, was developed and approved for chronic obesity. Both these branded medications were introduced to the Indian market prior to the patent's expiration.
Market Dynamics Evolve
The influx of nearly 50 new semaglutide brands is expected to lead to a concentrated market share, with a select few larger players likely to capture the majority of the demand. Industry observers anticipate that smaller companies' brands may struggle to gain traction, a pattern observed with previous patent-expired diabetes medications. Despite the heightened competition, the established market leaders, including the original patent holder and another major pharmaceutical firm, are unlikely to adjust their current pricing strategies. Their focus remains on their established clinical evidence, manufacturing processes, and extensive patient data. While newer generic options may gradually impact the market share of more expensive alternative drugs, such as tirzepatide, which has a later patent expiry, a swift shift is not anticipated. Patients already benefiting from these premium treatments may continue with them due to perceived quality and a desire to avoid potential initial side effects associated with switching. However, the introduction of significantly more affordable generic versions is projected to lead to a substantial increase, potentially two to three-fold, in the number of individuals utilizing these therapies, attracting those previously deterred by high costs.
Innovation in Delivery
The competitive edge in the newly expanded market isn't solely about price; it's also about innovation in drug delivery. Pharmaceutical companies are actively developing novel approaches to enhance patient convenience and treatment adherence. For instance, one leading company is preparing to launch semaglutide in multi-dose pens. These advanced pens will allow patients to precisely select and administer varying dose strengths from a single unit, which is particularly beneficial during the gradual dose escalation phase of treatment, often referred to as titration. This innovation contrasts with existing single-dose pens, which require patients to purchase different pens for each specific dosage increment. By simplifying the treatment process and offering greater flexibility, these new delivery mechanisms aim not only to improve the overall standard of care but also to encourage better long-term patient compliance with their prescribed regimen.
Navigating Misuse Risks
The burgeoning popularity of these weight loss medications, amplified by celebrity endorsements during the initial global launches, presents a tangible risk of misuse. A noticeable increase in marketing efforts by pharmaceutical companies, targeting healthcare professionals and initiating 'awareness campaigns,' is already evident. In response to potential promotional overreach, the country's top drug regulatory authority has issued advisories cautioning manufacturers against such advertising practices, emphasizing that any promotional activity, regardless of its pretext, that aims to create brand recall or product visibility for prescription drugs will be considered a violation. Furthermore, local chemist associations are reinforcing disciplinary measures, urging members to dispense these medications strictly based on doctor's prescriptions, especially in light of past instances where certain drugs were reportedly sold without proper prescriptions, a concern expected to escalate with a wider array of available medications. Healthcare professionals strongly advocate against self-medication and any form of misuse, stressing the importance of distinguishing between medical obesity management and cosmetic weight loss, and ensuring all treatments are administered under appropriate medical supervision to mitigate adverse effects, which can range from gastrointestinal issues to more serious complications like pancreatitis or thyroid concerns.