Regulatory Green Light
Alkem Laboratories has successfully obtained the necessary authorization from the Drug Controller General of India (DCGI) to manufacture and market its
Semaglutide Injection. This pivotal approval follows a thorough review of the company's Phase 3 clinical trials conducted within India, confirming the efficacy of the medication for its intended uses. The drug is specifically approved as an adjunct therapy for individuals managing type 2 diabetes mellitus and for those dealing with chronic weight management challenges, when used in conjunction with dietary modifications and regular exercise. This regulatory milestone marks a significant advancement in making advanced treatment options more accessible to the Indian population facing these prevalent health conditions.
Innovative Drug Delivery
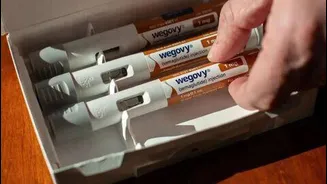
To enhance patient convenience and adherence, Alkem Laboratories is introducing Semaglutide in a pre-filled disposable injection pen format. This innovative delivery system is priced starting at approximately Rs 1,800 for a month's supply, translating to a weekly cost of around Rs 450. This approach simplifies the self-administration process for patients. Furthermore, for individuals requiring higher maintenance doses, the company also offers a reusable injection pen option. This dual approach caters to diverse patient needs and preferences, ensuring a more personalized and accessible treatment experience for those utilizing this new therapeutic agent for diabetes and weight management.
Brand Introductions
The newly approved Semaglutide injection is being launched in the Indian market under three distinct brand names: “Semasize”, “Obesema”, and “Hepaglide”. These varied brand names are intended to reach different patient segments and healthcare provider preferences, ensuring broad market penetration. Each brand signifies Alkem Laboratories' commitment to providing effective solutions for critical health concerns like type 2 diabetes and obesity. The introduction of these brands aims to raise awareness and offer tangible treatment options that can contribute to improved health outcomes for a significant number of patients across the nation.
Manufacturing Excellence
Underscoring its commitment to quality and global standards, Alkem Laboratories' manufacturing facility in Daman has received a 'Certificate of GMP Compliance of a Manufacturer' from the Malta Medicines Authority. This prestigious certification is valid for three years, concluding on December 9, 2025. Adding to its international accreditations, the company's manufacturing site in Baddi, India, has also been issued a 'Certificate of GMP Compliance of a Manufacturer' by the Department of Pharmacy (Human Medicines) – Germany, also valid for three years. These accreditations highlight Alkem's adherence to stringent Good Manufacturing Practices, ensuring the production of high-quality pharmaceuticals for both domestic and international markets.