What is the story about?
Drug firm, AstraZeneca Pharma India Ltd, on Friday (April 10), said it has received permission from the Central Drugs Standard Control Organisation (CDSCO) to import acalabrutinib tablets 100 mg, marketed as Calquence, for an additional indication in India.
The approval was granted on April 9, 2026, by the CDSCO under the Directorate General of Health Services, Government of India.
Following the approval, acalabrutinib tablets 100 mg in combination with venetoclax with or without obinutuzumab is indicated for the treatment of patients with previously untreated chronic lymphocytic leukaemia (CLL) or small lymphocytic lymphoma (SLL).
Also Read: AstraZeneca Pharma India gets CDSCO nod to import and market datverzo breast cancer drug
The company said the permission enables the import of Calquence tablets for sale and distribution in India for the specified additional indication. AstraZeneca Pharma India added that the marketing of acalabrutinib tablets 100 mg in the country for the additional indication will be subject to the receipt of related statutory approvals, if any.
Last week, AstraZeneca Pharma India said it had received approval from the Central Drugs Standard Control Organisation to import and market its cancer drug durvalumab (Imfinzi) for an additional indication in India.
In an exchange filing, the company said the approval allows the use of durvalumab solution for infusion (120 mg/2.4 mL and 500 mg/10 mL) for the treatment of patients with unresectable hepatocellular carcinoma (uHCC) who have not received prior systemic therapy.
Also Read: AstraZeneca Pharma Q3 Results: Topline jumps on oncology, biopharma push; margin slips
Durvalumab is an immunotherapy drug used in cancer treatment, and the expanded indication targets liver cancer patients who are not eligible for surgical intervention.
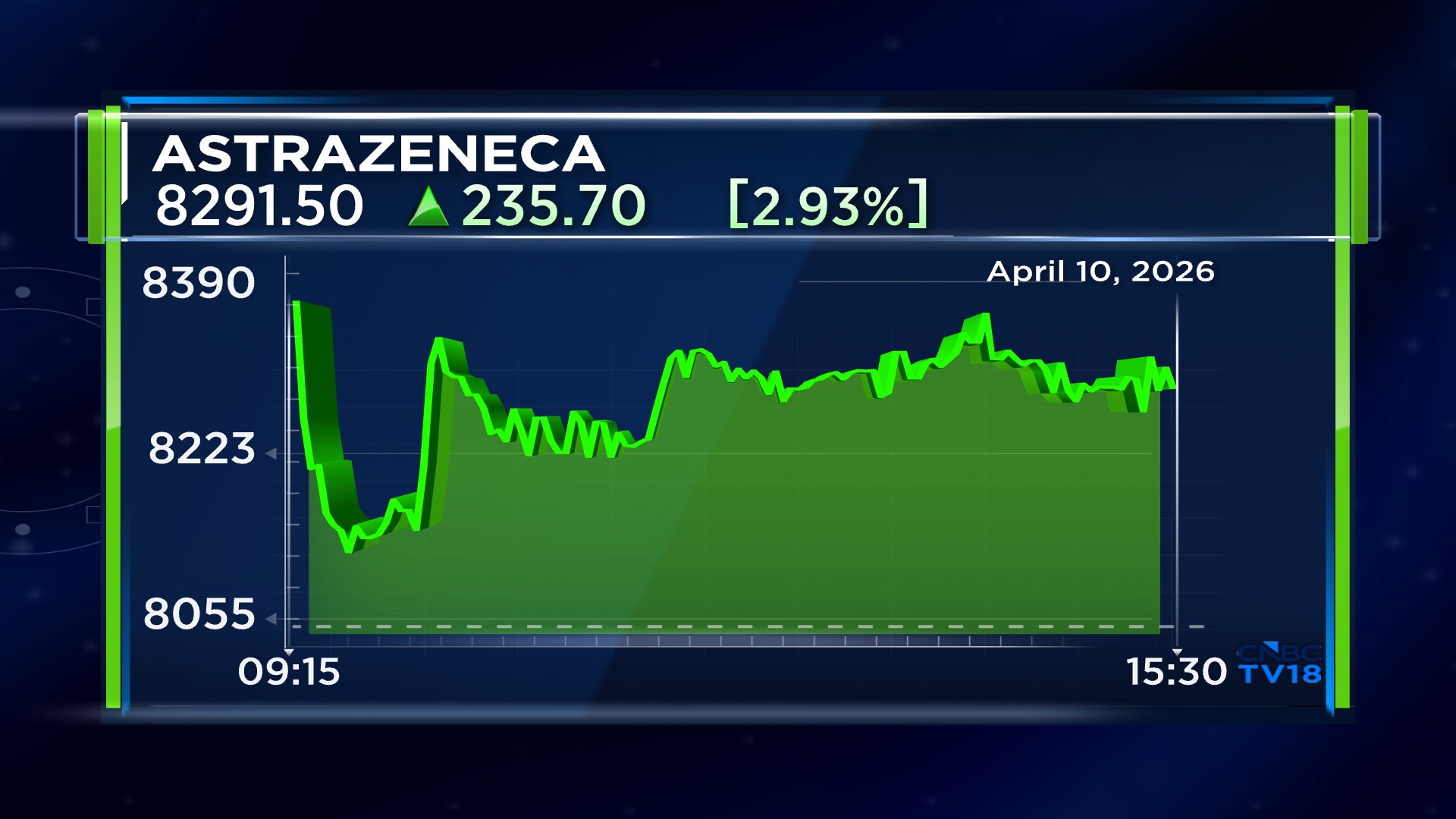
Shares of Astrazeneca Pharma India Ltd ended at ₹8,291.50, up by ₹235.70, or 2.93%, on the BSE today. April 10.
The approval was granted on April 9, 2026, by the CDSCO under the Directorate General of Health Services, Government of India.

Following the approval, acalabrutinib tablets 100 mg in combination with venetoclax with or without obinutuzumab is indicated for the treatment of patients with previously untreated chronic lymphocytic leukaemia (CLL) or small lymphocytic lymphoma (SLL).
Also Read: AstraZeneca Pharma India gets CDSCO nod to import and market datverzo breast cancer drug
The company said the permission enables the import of Calquence tablets for sale and distribution in India for the specified additional indication. AstraZeneca Pharma India added that the marketing of acalabrutinib tablets 100 mg in the country for the additional indication will be subject to the receipt of related statutory approvals, if any.
Last week, AstraZeneca Pharma India said it had received approval from the Central Drugs Standard Control Organisation to import and market its cancer drug durvalumab (Imfinzi) for an additional indication in India.
In an exchange filing, the company said the approval allows the use of durvalumab solution for infusion (120 mg/2.4 mL and 500 mg/10 mL) for the treatment of patients with unresectable hepatocellular carcinoma (uHCC) who have not received prior systemic therapy.
Also Read: AstraZeneca Pharma Q3 Results: Topline jumps on oncology, biopharma push; margin slips
Durvalumab is an immunotherapy drug used in cancer treatment, and the expanded indication targets liver cancer patients who are not eligible for surgical intervention.
Shares of Astrazeneca Pharma India Ltd ended at ₹8,291.50, up by ₹235.70, or 2.93%, on the BSE today. April 10.
/images/ppid_59c68470-image-177583255921550847.webp)

/images/ppid_59c68470-image-177564509378073281.webp)
/images/ppid_59c68470-image-177563015015136091.webp)
/images/ppid_59c68470-image-177565252740842963.webp)
/images/ppid_59c68470-image-177582752887567094.webp)
/images/ppid_59c68470-image-177583006584064472.webp)
/images/ppid_59c68470-image-177576004385551895.webp)
/images/ppid_59c68470-image-17758275303926994.webp)
/images/ppid_59c68470-image-177579253056980021.webp)
/images/ppid_59c68470-image-177575002903867742.webp)
/images/ppid_59c68470-image-177580002884489126.webp)
/images/ppid_59c68470-image-177565753436589709.webp)
/images/ppid_59c68470-image-177566253175645230.webp)