What's Happening?
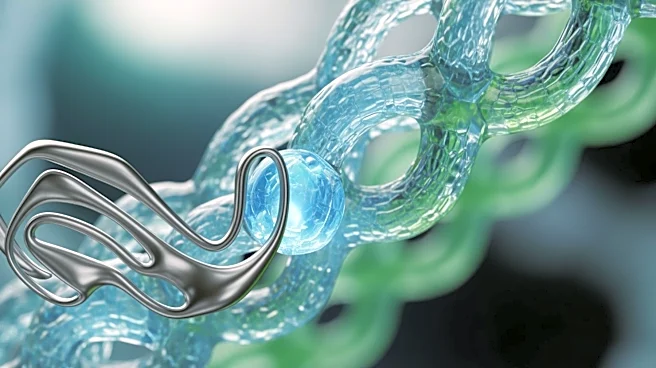
A team of international researchers, including experts from the University of Arkansas and Japanese institutions, has discovered the molecular mechanism by which bacterial collagenase breaks down collagen. This enzyme, known as ColH, engages with collagen at the atomic
level to catalyze its cleavage, a process crucial for bacterial tissue invasion. The study, published in Nature Communications, reveals how the enzyme's dynamic conformational shifts enable it to degrade collagen efficiently. This understanding could lead to the development of improved therapeutic enzymes for use in transplantation and regenerative medicine. The research highlights the enzyme's potential to enhance cancer treatments by stripping away collagen shields that protect tumors, thereby increasing the efficacy of chemotherapeutic agents.
Why It's Important?
The discovery of the bacterial collagenase mechanism has significant implications for medical science. By understanding how this enzyme degrades collagen, researchers can design tailored enzymes with enhanced specificity and efficiency. Such enzymes could revolutionize treatments for fibrosis, improve transplantation techniques, and aid in cancer therapy by making tumors more susceptible to treatment. The study also underscores the potential for bioengineering applications, where these enzymes could be used to develop new therapeutic strategies. This research not only advances our knowledge of bacterial virulence but also opens new avenues for medical innovation, highlighting the intersection of evolutionary biology and therapeutic development.