What's Happening?
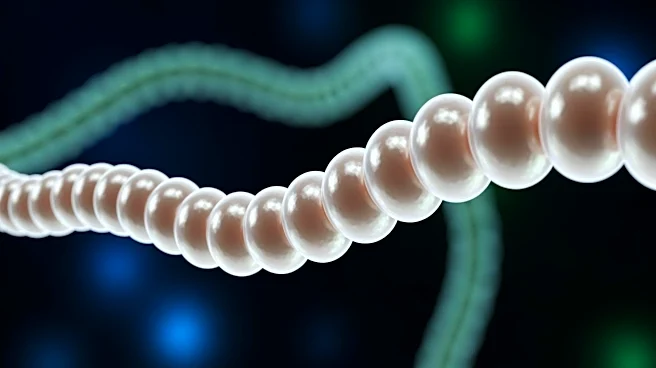
Researchers at the Laboratory of Experimental Biophysics (LEB) at EPFL have discovered a new mechanism called 'mitochondrial pearling' that plays a crucial role in the organization of mitochondrial DNA (mtDNA) within cells. Mitochondria, known as the powerhouses
of the cell, contain their own genetic material, which is organized into structures called nucleoids. These nucleoids are evenly spaced within mitochondria, a pattern essential for proper cellular function. The study, led by Professor Suliana Manley and postdoctoral fellow Juan Landoni, reveals that mitochondrial pearling involves temporary shape changes in mitochondria, forming bead-like structures that help redistribute mtDNA evenly. This process is triggered by calcium influx and is crucial for maintaining nucleoid spacing, which is vital for cellular health and function.
Why It's Important?
The discovery of mitochondrial pearling provides significant insights into how cells maintain their internal organization, which is critical for their function and survival. Proper spacing of mtDNA is essential for the expression of mitochondrial genes and the overall energy production of the cell. Disruptions in this process have been linked to various metabolic and neurological disorders, including Alzheimer's and Parkinson's diseases. Understanding mitochondrial pearling could lead to new therapeutic strategies for these conditions by targeting the mechanisms that regulate mtDNA distribution. This research highlights the importance of physical processes in cellular organization, offering a new perspective on mitochondrial biology and its implications for human health.
What's Next?
Future research will likely focus on further elucidating the molecular mechanisms that regulate mitochondrial pearling and its role in disease. Scientists may explore potential therapeutic interventions that can modulate this process to treat or prevent diseases associated with mitochondrial dysfunction. Additionally, the study opens avenues for investigating other physical processes that contribute to cellular organization and their impact on health and disease. The findings could also inspire new approaches in biotechnology and synthetic biology, where controlled manipulation of cellular structures is crucial.