What's Happening?
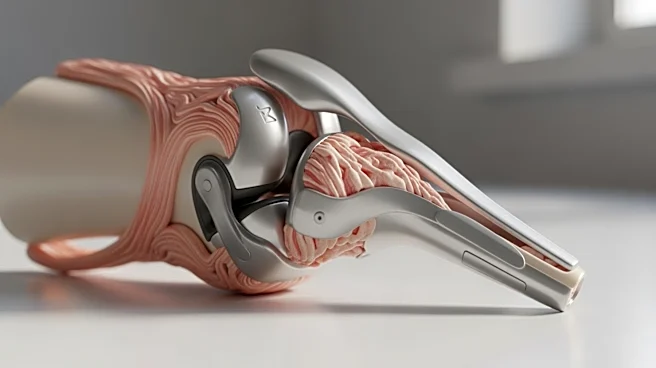
Researchers at Columbia University and the University of Missouri are developing a new type of knee implant called NOVAKnee, which is made from biodegradable materials and stem-cell-derived bone and cartilage. This 'living' knee replacement aims to provide
a longer-lasting solution for patients, particularly younger individuals who face limitations with conventional metal and plastic implants. Traditional knee replacements typically last 15 to 20 years, often necessitating revision surgeries for younger patients. The NOVAKnee is designed to integrate with the patient's body, gradually replacing the scaffold with natural bone and cartilage. The implant has been tested in lab mice and will soon undergo trials in larger animals, with human trials anticipated by 2028. This development is part of a federally funded project focused on tissue regeneration in osteoarthritis.
Why It's Important?
The development of the NOVAKnee represents a significant advancement in orthopedic surgery, particularly for younger patients who currently have limited options for knee replacements. Conventional implants often require revision surgeries, which can be complex and risky, especially as patients age. By potentially offering a longer-lasting and more natural solution, the NOVAKnee could reduce the need for multiple surgeries and improve the quality of life for patients. This innovation also highlights the growing field of regenerative medicine, which seeks to harness the body's natural healing processes to repair and replace damaged tissues. If successful, this technology could pave the way for similar advancements in other joint replacements, addressing a broader range of orthopedic issues.
What's Next?
The next steps for the NOVAKnee project include conducting large animal trials to better simulate human knee replacement conditions. These trials will help determine the implant's effectiveness and safety before moving to human trials, which are expected to begin by 2028. Researchers are also exploring the potential for using the technology in other joints, such as the thumb, which could expand the application of this regenerative approach. As the project progresses, it will be crucial to address any technical challenges and refine the clinical workflow to ensure the best outcomes for patients. The success of these trials could lead to a paradigm shift in how joint replacements are approached, with a focus on regenerative solutions.
Beyond the Headlines
The development of the NOVAKnee raises important ethical and practical considerations. The use of stem cells, whether autologous or allogeneic, involves complex decisions about sourcing and patient suitability. Additionally, the integration of living materials into medical implants challenges traditional notions of medical devices, potentially leading to new regulatory and clinical practices. This innovation also underscores the importance of interdisciplinary collaboration in advancing medical technology, combining expertise from biomedical engineering, orthopedic surgery, and regenerative medicine. As the technology evolves, it may also influence public perceptions of medical implants and the role of biotechnology in healthcare.