What's Happening?
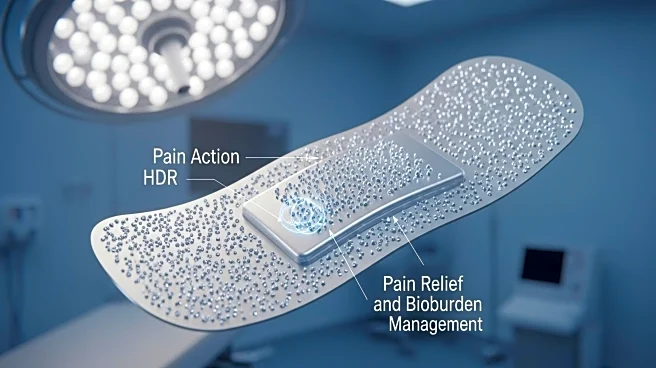
Imbed Biosciences Inc., a company specializing in advanced wound care technology, has introduced SAM™ PainGuard™ with Lidocaine, a new addition to its portfolio of synthetic, antimicrobial, and biocompatible wound matrices. This product launch includes Surgaflex™ PainGuard™, designed for surgical wounds, and Pelashield™ PainGuard™, aimed at managing burn and trauma wounds. The innovation integrates multiple wound care agents into a single matrix composed of bioresorbable polymers, providing both antimicrobial protection and acute pain relief. The matrix delivers a controlled release of lidocaine HCl, offering immediate pain relief and is indicated for various wound types, including surgical, burn, donor site, trauma, and chronic wounds.
Why It's Important?
The introduction of SAM™ PainGuard™ is significant as it addresses the dual challenges of managing bioburden and pain in wound care, potentially reducing reliance on systemic pain medications, including opioids. This is crucial for improving patient outcomes, particularly in burn care, where pain management is critical to prevent long-term complications such as anxiety, PTSD, and delayed healing. The product's ability to provide immediate pain relief and sustained microbial reduction can streamline clinical workflows and enhance patient care, offering a convenient solution for clinicians and reducing possible side effects associated with uneven lidocaine applications.
What's Next?
Imbed Biosciences plans to expand its portfolio of SAM™-based wound care solutions, with Surgaflex™ PainGuard™ and Pelashield™ PainGuard™ now available in the U.S. The company aims to continue developing clinically relevant solutions that simplify clinical workflows and improve patient outcomes. The FDA 510(k)-cleared product is expected to gain traction among healthcare providers, potentially leading to broader adoption in various medical settings.